Aleo BME receives FDA Clearance for its ElaSkinTM Liquid Bandage
The First Commercial Product from Aleo BME’s Bioactive Polymer Platform
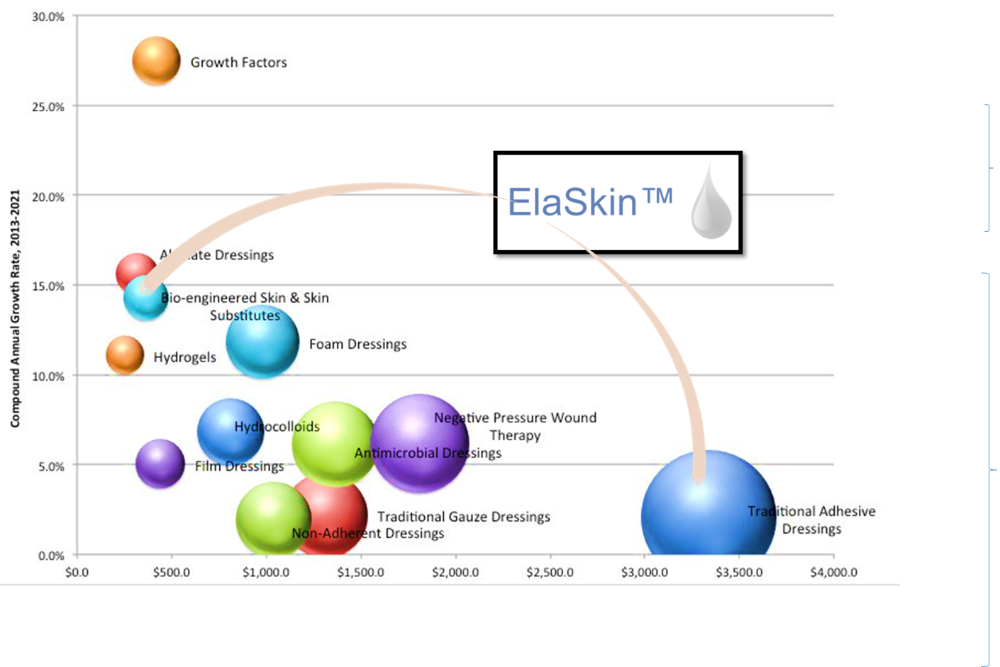
State College, PA, January 2018- Biomaterials and medical device start-up, Aleo BME, has received notification from the U.S. FDA that it has been approved for the sale and licensing of ElaSkinTM as a liquid bandage for the protection and treatment of a broad set of skin conditions and injuries. The 510K clearance introduces the market to a dynamic product capable of partly addressing the $10B global wound care market and is the first product to come from Aleo BME’s growing platform of bioactive polymers being developed for in health care, cosmetic, and agricultural applications.
ElaSkin’s development began in 2016 and validates Aleo BME’s approach to biopolymer production and characterization and demonstrates an ability to enter critical, regulated markets and produce medical devices that can benefit large patient populations. Aleo BME’s development pipeline also includes medical devices for unmet needs in the fields of endoscopy and neurology, enabling biopolymeric solutions that recognize the unique biology of diverse tissues. Offering to put the 510K clearance in context, CEO Chao Liu says “ElaSkinTM is a best-in-class technology that has a lot of potential uses in health care beyond the growing liquid bandage market. Its structure and strength afford it a degree of comfort and durability that no other marketed, biocompatible material has achieved. We’ve found a lot of uses for this science, but the breadth and growth of the wound care market makes it a great place to start.”
Aleo BME’s foundational technology is comprised of novel biopolymers and nanoparticles developed within its medical device R&D program. Aleo BME is currently partnering for scale-up and distribution of the ElaSkinTM liquid bandage and expects to make it available for both prescription and over-the-counter use by 2019.
-lw-scaled.png.png)